08.05.2026
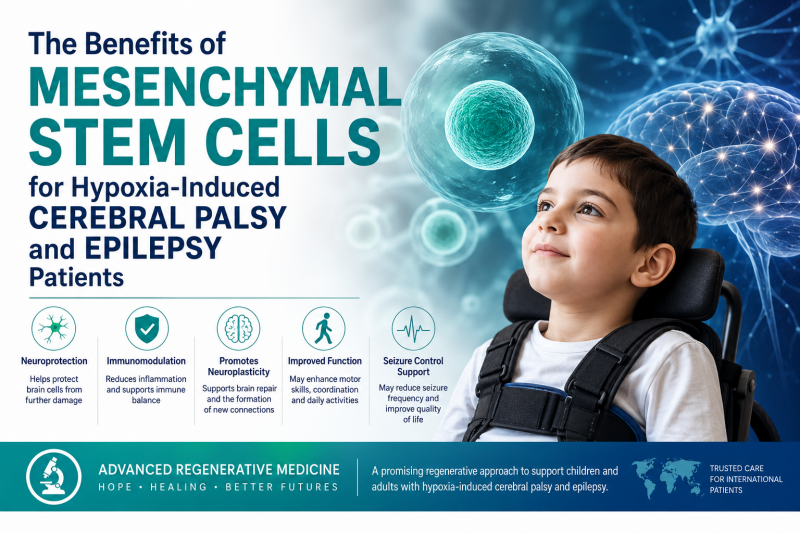
Benefits of Mesenchymal Stem Cells in Hypoxia-Related Cerebral Palsy and Epilepsy
Advanced Regenerative Treatment Options for International Patients Seeking Cerebral Palsy and Epilepsy Care in Turkey
Introduction
Hypoxia-related cerebral palsy and epilepsy are complex neurological conditions that often begin with oxygen deprivation to the brain before, during, or shortly after birth. This type of brain injury may lead to long-term motor impairment, developmental delay, muscle stiffness, coordination problems, speech delay, cognitive challenges, feeding difficulties, and seizures.
For many families, especially those from the USA, Canada, CIS countries, the Balkans, and Germany, standard treatment options may feel limited. Traditional care usually focuses on rehabilitation, anti-seizure medications, orthopedic management, and supportive therapies. These remain essential. However, in recent years, mesenchymal stem cells, also called mesenchymal stromal cells or MSCs, have gained increasing scientific attention as a potential regenerative and immunomodulatory therapy for neurological injury.
MSC therapy is not a miracle cure and should not be presented as one. However, growing clinical and preclinical research suggests that MSCs may help support brain repair mechanisms, reduce inflammation, improve the neurological environment, and contribute to functional gains in selected patients with cerebral palsy and certain epilepsy-related neurological injuries. Current evidence is promising but still evolving, and international medical guidance emphasizes the importance of proper patient selection, ethical treatment protocols, and realistic expectations.
Understanding Hypoxia-Related Cerebral Palsy
Cerebral palsy is a group of permanent movement and posture disorders caused by injury to the developing brain. One of the important causes is hypoxic-ischemic injury, where the brain receives insufficient oxygen and blood flow. This may occur because of birth complications, placental problems, neonatal respiratory distress, infection, premature birth, prolonged labor, or other perinatal complications.
When oxygen deprivation damages the developing brain, the injury may affect areas responsible for movement, muscle tone, coordination, balance, speech, cognition, and seizure control. Hypoxic-ischemic encephalopathy, commonly known as HIE, is recognized as a major cause of long-term neurological disability, including cerebral palsy and epilepsy.
Children with hypoxia-related cerebral palsy may present with:
• Spasticity or increased muscle tone
• Delayed motor milestones
• Poor head control, sitting, crawling, or walking delay
• Abnormal reflexes
• Speech and swallowing difficulties
• Learning or cognitive delay
• Epileptic seizures
• Visual or hearing problems
• Feeding and growth difficulties
• Orthopedic deformities over time
The severity depends on the timing, location, and extent of the brain injury.
Why Epilepsy Can Occur After Hypoxic Brain Injury
Epilepsy may develop when injured brain tissue creates abnormal electrical activity. In children with hypoxic-ischemic brain injury, seizures may begin during the neonatal period or appear later in infancy or childhood. Some seizures may resolve, while others may become recurrent and require long-term anti-seizure medication. Epilepsy.com explains that HIE can cause brain injury that may lead to seizures, and in some cases seizures may recur weeks to years later.
The International League Against Epilepsy also notes that seizure burden in hypoxic-ischemic encephalopathy is often a marker of the extent of brain injury, meaning that epilepsy management must be integrated with full neurological rehabilitation and developmental care.
For this reason, patients with cerebral palsy and epilepsy require a multidisciplinary approach, including pediatric neurology, physical rehabilitation, neuroimaging, EEG monitoring, anti-seizure medication optimization, and, in selected cases, regenerative medicine evaluation.
What Are Mesenchymal Stem Cells?
Mesenchymal stem cells, more accurately referred to in many scientific publications as mesenchymal stromal cells, are specialized cells that can be isolated from tissues such as:
• Umbilical cord tissue
• Umbilical cord blood
• Bone marrow
• Adipose tissue
• Placental tissue
In neurological conditions, MSCs are not mainly expected to “replace” damaged brain cells directly. Instead, their potential benefit appears to come from their ability to release biologically active molecules, regulate inflammation, support tissue repair, modulate immune responses, and improve the microenvironment around injured cells. This is known as paracrine signaling.
A major 2025 scoping review and meta-analysis on MSC therapy for cerebral palsy reported that MSCs are commonly derived from umbilical cord tissue, bone marrow, and adipose tissue, and that their effects are believed to be largely related to anti-inflammatory, immunomodulatory, and paracrine mechanisms rather than direct cell replacement.
How MSCs May Help in Hypoxia-Related Cerebral Palsy
Hypoxic brain injury does not only create a one-time injury. In many patients, the brain may remain affected by chronic inflammation, altered immune activity, impaired myelination, oxidative stress, reduced neuroplasticity, and abnormal neural network development. MSCs are being studied because they may target several of these mechanisms at the same time.
1. Reducing Neuroinflammation
Inflammation plays an important role in brain injury after hypoxia. MSCs may help regulate inflammatory pathways and reduce harmful immune activity. Research suggests that MSCs can interact with immune cells and shift inflammatory responses toward a more balanced, anti-inflammatory state.
This may be especially important in children with cerebral palsy, where inflammation may remain relevant years after the original brain injury.
2. Supporting Neuroprotection
Preclinical studies in hypoxic-ischemic brain injury suggest that MSCs may help protect neurons, reduce cell death, and support recovery in injured brain tissue. A PLOS One systematic review of preclinical studies described MSCs as promising therapeutic agents in experimental models of neonatal hypoxic-ischemic encephalopathy.
Animal studies of umbilical cord-derived MSCs have also shown neuroprotective effects after hypoxic-ischemic injury, including reduced brain inflammation and improved neuronal survival.
3. Improving the Brain Repair Environment
MSCs release cytokines, growth factors, extracellular vesicles, and other signaling molecules that may support tissue repair, vascular health, myelination, and neuroplasticity. In cerebral palsy research, these effects are considered highly relevant because functional improvement often depends on the brain’s ability to reorganize and strengthen remaining neural pathways.
4. Potential Improvement in Gross Motor Function
One of the most important clinical outcomes in cerebral palsy is gross motor function, often measured by the Gross Motor Function Measure, or GMFM. A 2025 scoping review and meta-analysis identified 30 published reports and 10 registered trials, including 1,292 people with cerebral palsy who received MSCs. The review reported that MSC treatment was generally safe and that meta-analysis showed significant improvements in GMFM outcomes at 3, 6, and 12 months after treatment. However, the authors also emphasized that study heterogeneity and the lack of published Phase 3 trials limit definitive conclusions.
This means MSC therapy may be a promising supportive option for motor function, but it should be integrated with intensive rehabilitation rather than viewed as a standalone treatment.
5. Supporting Rehabilitation Response
In practical clinical care, MSC therapy is usually considered most valuable when combined with structured rehabilitation. The goal is not only to administer cells but also to create a period in which the patient’s nervous system may respond better to physiotherapy, occupational therapy, speech therapy, balance training, and neurodevelopmental exercises.
For international patients, this is an important point: the best programs are not “injection-only” approaches. They are multidisciplinary neurological rehabilitation programs where regenerative medicine is combined with careful assessment, therapy planning, and follow-up.
Potential Benefits of MSC Therapy for Cerebral Palsy Patients
Based on current research and clinical experience, possible benefits may include:
• Improved gross motor function
• Better muscle tone regulation
• Reduced spasticity in selected patients
• Improved posture and balance
• Better coordination
• Improved hand function in some cases
• Better rehabilitation response
• Possible improvement in activities of daily living
• Potential improvement in cognition, communication, or social interaction in selected patients
• Better quality of life when combined with rehabilitation
A 2024 evidence brief from the Agency for Clinical Innovation in New South Wales summarized that recent trials have shown promising safety and efficacy profiles for cell-based therapies in cerebral palsy. It also noted that patients receiving human umbilical cord mesenchymal stem cells in a randomized controlled trial had significant improvements in activities of daily living, comprehensive function assessment, and gross motor function at 12-month follow-up, with similar safety profiles between treatment and control groups.
MSC Therapy and Epilepsy: What Does the Evidence Show?
The evidence for MSC therapy in epilepsy is more preliminary than the evidence for cerebral palsy motor outcomes. Epilepsy is not one single disease; it can result from structural brain injury, genetic causes, metabolic disease, malformations, infection, or unknown factors. Therefore, patient selection is very important.
In hypoxia-related cerebral palsy, epilepsy may be associated with scarred or damaged brain networks. MSCs are being studied because they may reduce neuroinflammation, support neural repair mechanisms, and potentially influence the abnormal environment that contributes to seizure activity.
A systematic review on MSC therapy for focal epilepsy reported that MSCs have been evaluated in preclinical and early clinical studies for drug-resistant epilepsy. A 2024 review also concluded that MSCs hold promise in epilepsy because of their anti-inflammatory and neuroprotective properties, while emphasizing the need for further investigation.
For patients, this means MSC therapy should not be presented as a replacement for anti-seizure medications, EEG monitoring, epilepsy surgery evaluation, or standard neurological care. Instead, it may be considered as a supportive regenerative approach in selected patients, especially when epilepsy is linked to structural hypoxic brain injury and chronic neuroinflammation.
Can MSC Therapy Stop Seizures?
At this stage, it would not be medically responsible to promise that MSC therapy will stop seizures. Some early studies and case-based research suggest possible seizure reduction in selected epilepsy patients, but the evidence remains limited and not yet strong enough to replace established epilepsy treatments. ClinicalTrials.gov lists studies investigating MSCs for drug-resistant epilepsy, reflecting ongoing scientific interest, but these are still investigational approaches rather than universally accepted standard epilepsy therapy.
The most realistic goals for epilepsy patients may include:
• Supporting overall neurological function
• Reducing neuroinflammatory burden
• Improving developmental and rehabilitation potential
• Possibly reducing seizure burden in selected patients
• Improving quality of life when combined with optimized epilepsy management
All anti-seizure medications must be managed by a neurologist. Medication changes should never be made independently after MSC therapy.
Why Umbilical Cord-Derived MSCs Are Commonly Used
Umbilical cord-derived MSCs are widely studied because they are young, biologically active, ethically obtainable after healthy births, and suitable for allogeneic use under proper laboratory standards. They are also considered to have low immunogenicity, which may allow repeat dosing in selected protocols.
In cerebral palsy research, umbilical cord tissue-derived MSCs and umbilical cord blood-based cell therapies are among the most studied approaches. The 2025 MSC cerebral palsy review found that most patients in the reviewed studies received umbilical cord tissue MSCs, commonly through intrathecal or intravenous routes.
Routes of MSC Administration
The route of administration must be selected by the medical team according to the patient’s diagnosis, age, neurological status, MRI findings, seizure control, general health, and risk profile.
Common routes studied in neurological conditions include:
- Intrathecal Administration
Intrathecal administration delivers MSCs into the cerebrospinal fluid through a lumbar puncture. This route is often considered in neurological conditions because it places cells closer to the central nervous system environment.
Potential temporary side effects may include headache, fever, irritability, nausea, vomiting, low back discomfort, or symptoms related to cerebrospinal fluid pressure changes. In the 2025 cerebral palsy MSC review, reported intrathecal safety events were generally treatable and resolved within days.
- Intravenous Administration
Intravenous administration delivers MSCs through the bloodstream. It is less invasive than intrathecal delivery but may have different biodistribution. Systemic administration may not result in large numbers of MSCs crossing the blood-brain barrier; however, immunomodulatory effects may still occur through systemic immune signaling.
Temporary infusion-related reactions such as fever, vomiting, diarrhea, or breathing discomfort have been reported in some studies, usually mild and transient.
- Combined Protocols
Some centers use combined or repeated protocols depending on patient selection and medical indications. However, optimal dose, route, frequency, and timing have not yet been definitively established by Phase 3 trials. This is why treatment must be individualized and carefully explained to families.
Who May Be a Candidate for MSC Therapy?
MSC therapy may be considered for selected patients with:
• Hypoxia-related cerebral palsy
• Spastic cerebral palsy
• Mixed-type cerebral palsy
• Cerebral palsy with developmental delay
• Cerebral palsy with controlled or partially controlled epilepsy
• Brain injury after hypoxic-ischemic encephalopathy
• Motor delay associated with perinatal brain injury
• Patients already receiving rehabilitation but seeking additional supportive options
However, candidacy must be determined after detailed review.
A proper pre-treatment evaluation may include:
• Brain MRI review
• EEG or video EEG if seizures are present
• Pediatric neurology or adult neurology consultation
• Neurosurgery or regenerative medicine consultation when intrathecal treatment is considered
• Physical medicine and rehabilitation assessment
• Review of anti-seizure medications
• Blood tests and infection screening
• Immune and metabolic evaluation when needed
• Functional scoring such as GMFM or developmental assessment
• Review of previous therapies and rehabilitation history
Patients with uncontrolled infection, unstable epilepsy, severe systemic illness, active malignancy, serious immune disorders, or unsafe anesthesia/sedation risk may not be suitable.
Safety and Ethical Considerations
MSC therapy has shown a generally favorable safety profile in many cerebral palsy studies, but this does not mean it is risk-free. The field remains under active investigation, and families should avoid clinics that promise guaranteed cures, rapid reversal of cerebral palsy, or complete seizure elimination.
The U.S. FDA states that regenerative medicine therapies have not been approved to treat neurological disorders such as epilepsy or stroke, and that currently FDA-approved stem cell products are limited to certain blood-forming stem cells from umbilical cord blood for specific hematologic conditions.
A 2024 evidence brief also noted that, internationally, no jurisdictions had approved stem cell therapies for cerebral palsy at that time, and that most clinical trials were Phase I or Phase II, with a lack of Phase III trials required for regulatory approval.
For international patients, this information is especially important. A professional center should provide:
• Transparent explanation of evidence
• Clear informed consent
• Realistic expectations
• Laboratory quality documentation
• Sterility and safety standards
• Physician-led treatment planning
• Neurological follow-up
• Integration with rehabilitation
• No exaggerated promises
Why International Patients Travel to Turkey for Advanced Neurological and Regenerative Care
Turkey has become an important destination for international patients seeking advanced neurological evaluation, rehabilitation, and regenerative medicine options. Istanbul is especially attractive because it combines experienced physicians, internationally oriented hospitals, multilingual medical coordination, and easier access for patients from Europe, CIS countries, the Middle East, and the Balkans.
Patients from the USA and Canada often look for options when access to investigational regenerative treatments is limited or difficult. Patients from Germany and other German-speaking countries usually prioritize medical quality, documentation, safety, and transparent physician communication. Patients from the CIS region and Balkans often seek faster access to advanced diagnostics, second opinions, and coordinated treatment abroad.
For these patients, the most important factor is not only the availability of MSC therapy, but the quality of the entire treatment pathway.
RecMed’s Approach to Hypoxia-Related Cerebral Palsy and Epilepsy Patients
At RecMed Medical Travel, patients are evaluated through a multidisciplinary and medically responsible pathway. The goal is to understand the patient’s neurological condition in detail before recommending any regenerative treatment.
A typical evaluation pathway may include:
1. Medical document review
Previous MRI reports, EEG results, epilepsy history, birth history, rehabilitation records, medication lists, and videos of motor function or seizures may be reviewed.
2. Neurological assessment
A pediatric neurologist or neurologist evaluates seizure control, developmental status, motor function, and current medication strategy.
3. MRI and EEG-based planning
Updated MRI, EEG, or video EEG may be recommended to better understand the injury pattern and seizure activity.
4. Regenerative medicine suitability review
The patient’s candidacy for MSC therapy is assessed according to safety, diagnosis, functional status, and realistic expected benefit.
5. Rehabilitation integration
MSC therapy is ideally combined with physiotherapy, occupational therapy, speech therapy, and neurodevelopmental rehabilitation.
6. Post-treatment follow-up
Progress is monitored over time, because functional changes may appear gradually over weeks or months.
Expected Outcomes: What Families Should Realistically Understand
The goal of MSC therapy in hypoxia-related cerebral palsy is not to “cure” cerebral palsy. Cerebral palsy is caused by permanent injury to the developing brain. However, the brain may still have capacity for adaptation, especially in children. MSC therapy aims to support the biological environment for recovery and enhance rehabilitation potential.
Possible realistic outcomes may include:
• Better muscle control
• Reduced stiffness
• Improved sitting, standing, or walking capacity in selected patients
• Improved hand use
• Better balance
• Improved endurance
• Better interaction and alertness
• Improved rehabilitation response
• Possible reduction in seizure frequency in selected epilepsy patients
• Improved quality of life
The degree of improvement varies significantly. Factors that influence outcomes include age, severity of brain injury, type of cerebral palsy, seizure control, MRI findings, baseline motor function, rehabilitation intensity, nutrition, orthopedic status, and overall medical condition.
Why Rehabilitation Remains Essential After MSC Therapy
MSC therapy should never replace rehabilitation. In fact, rehabilitation may be one of the most important factors that determines whether biological changes can translate into functional improvement.
After MSC therapy, patients may benefit from:
• Intensive physiotherapy
• Occupational therapy
• Speech and swallowing therapy
• Balance and coordination training
• Robotic rehabilitation when appropriate
• Spasticity management
• Orthopedic follow-up
• Nutrition support
• Family-guided home exercises
The most successful programs are usually those that combine regenerative medicine with structured neurological rehabilitation.
Important Questions Families Should Ask Before Choosing MSC Therapy
International patients should ask every clinic or medical travel provider the following questions:
• What type of MSCs are used?
• What is the source of the cells?
• Are the cells tested for sterility and safety?
• Is the treatment physician-led?
• Is there a neurologist involved?
• Is EEG evaluation performed for epilepsy patients?
• What route of administration is recommended and why?
• What are the possible side effects?
• What benefits are realistic in this specific case?
• Is rehabilitation included in the treatment plan?
• How will progress be measured?
• What follow-up is provided after returning home?
A trustworthy center should answer these questions clearly and should not pressure families with unrealistic promises.
Conclusion
Mesenchymal stem cell therapy is one of the most promising areas of regenerative medicine for hypoxia-related cerebral palsy and selected epilepsy-related neurological injuries. Current evidence suggests that MSCs may help through anti-inflammatory, immunomodulatory, neuroprotective, and paracrine mechanisms. Clinical studies in cerebral palsy have reported encouraging safety data and potential improvements in gross motor function, especially when treatment is combined with rehabilitation.
However, MSC therapy remains an evolving medical field. It should be offered only with careful patient selection, transparent informed consent, realistic expectations, and specialist neurological supervision. For epilepsy patients, MSC therapy should be considered supportive and investigational, not a replacement for established anti-seizure treatment.
For international families seeking advanced options beyond standard supportive care, Turkey offers a strong medical travel pathway when regenerative medicine is combined with neurology, rehabilitation, diagnostics, and long-term follow-up.
References
1. Paton MCB, Griffin AR, Blatch-Williams R, et al. Clinical Evidence of Mesenchymal Stromal Cells for Cerebral Palsy: Scoping Review with Meta-Analysis of Efficacy in Gross Motor Outcomes. Cells. 2025.
2. Agency for Clinical Innovation, NSW. Evidence Brief: Umbilical Cord Blood Therapy for Cerebral Palsy. 2024.
3. U.S. Food & Drug Administration. Important Patient and Consumer Information About Regenerative Medicine Therapies.
4. U.S. Food & Drug Administration. Consumer Alert on Regenerative Medicine Products Including Stem Cells and Exosomes.
5. Archambault J, Moreira A, McDaniel D, et al. Therapeutic Potential of Mesenchymal Stromal Cells for Hypoxic Ischemic Encephalopathy: A Systematic Review and Meta-Analysis of Preclinical Studies. PLOS One. 2017.
6. McDonald CA, et al. Intranasal Delivery of Mesenchymal Stromal Cells Protects Against Neonatal Hypoxic-Ischemic Brain Injury. 2019.
7. Epilepsy Foundation. Hypoxic Ischemic Encephalopathy and Seizures.
8. International League Against Epilepsy. Treatment of Seizures in the Neonate: Guidelines and Consensus-Based Recommendations.
9. Ramos-Fresnedo A, et al. Mesenchymal Stem Cell Therapy for Focal Epilepsy: A Systematic Review. Epilepsia. 2022.
10. Mattei V, et al. Mesenchymal Stem Cells and Their Role in Epilepsy. Cells. 2024.
11. ClinicalTrials.gov. Efficacy of Allogeneic Umbilical Cord Blood Infusion and MSCs in Cerebral Palsy.
12. ClinicalTrials.gov. Safety of Autologous MSC Infusion to Treat Epilepsy.